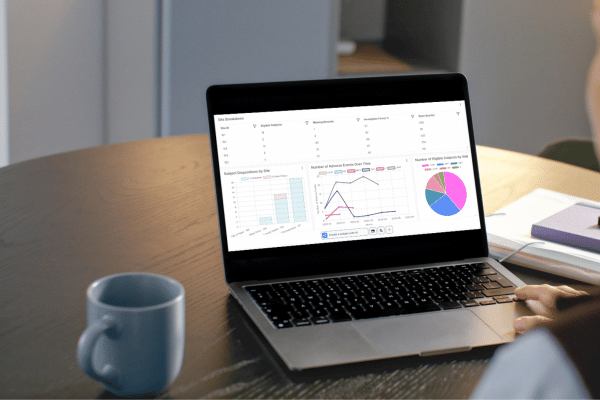
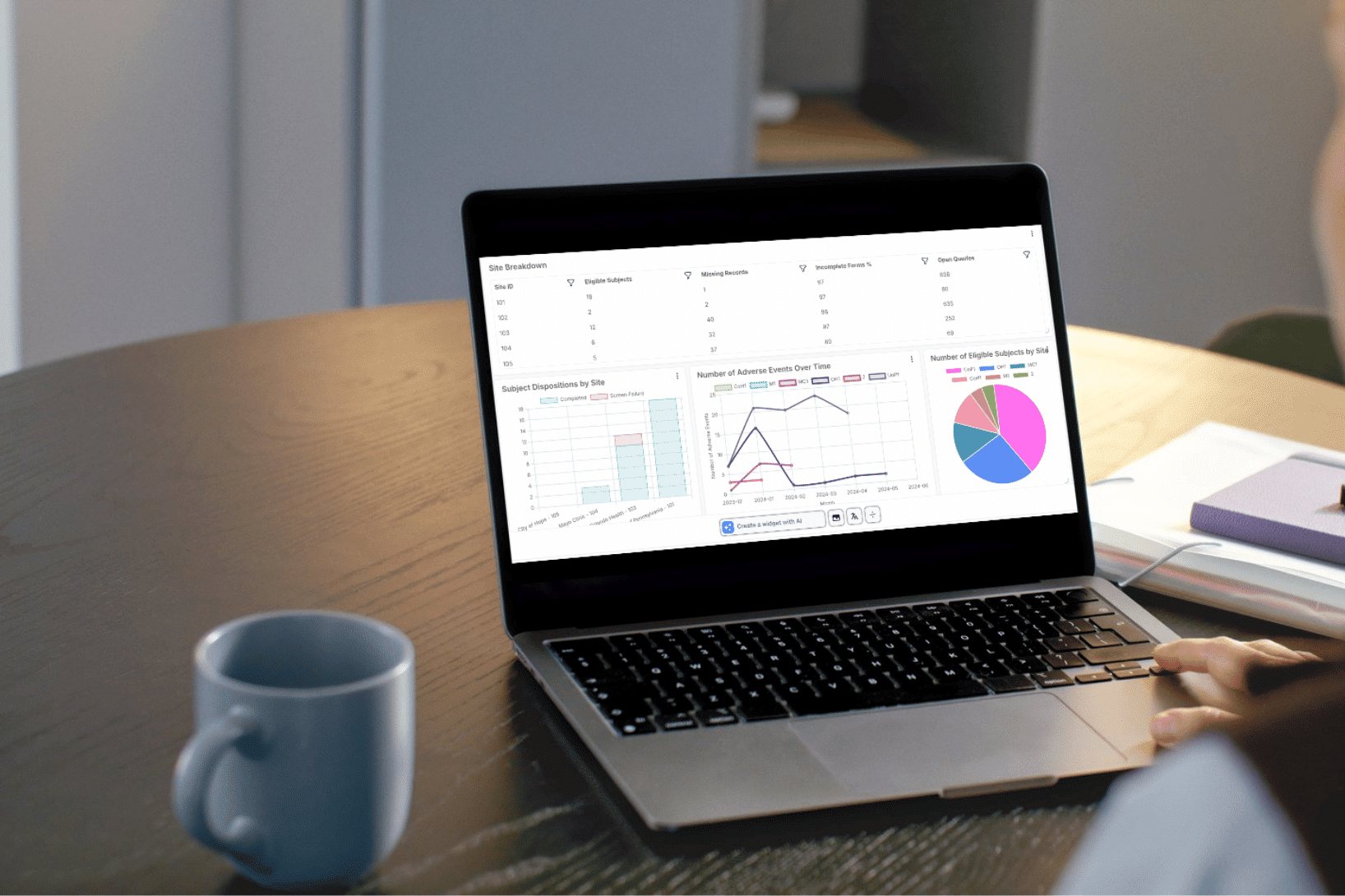
TrialKit AI: Intelligence for Clinical Trials
Design, build, simulate, and analyze clinical studies in one connected, insight-driven system. Embedded intelligence supports the full study lifecycle, from protocol development through study closeout.


What is TrialKit AI?
TrialKit AI is an embedded intelligence layer built directly into the TrialKit platform, extending across the full study lifecycle, supporting study design, configuration, validation, simulation, and analysis within the same environment.
Unlike standalone analytics tools or external AI systems, TrialKit AI operates within the same environment used to build and manage clinical studies. This allows data analysis, simulation, CRF generation, and validation to occur within the full context of the study’s protocol, forms, visits, and endpoints.
Powered by Floyd, the proprietary AI model developed by Crucial Data Solutions, TrialKit AI enables research teams to interact with their data using natural language, perform advanced statistical analysis, and simulate potential study outcomes before enrollment begins. These capabilities help sponsors and CROs move beyond traditional reporting toward a deeper understanding of how studies perform across their lifecycle.
Powered by Floyd
The Proprietary AI Model Behind TrialKit AI
Floyd is a domain-aware AI model tuned from Google Gemini and designed specifically for clinical research. Unlike general AI systems that analyze flattened datasets, Floyd understands the structural architecture of clinical trials, including protocol logic, visits, forms, endpoints, and longitudinal data relationships.
Floyd powers intelligence across each stage of the study lifecycle, from initial design decisions through ongoing analysis. Within TrialKit AI, Floyd powers four core capabilities:
- Floyd Study Simulation
- Floyd Validation
- Floyd Chat
- Floyd Analytics
Together, these capabilities transform how research teams explore, analyze, and guide clinical trials.
Floyd Study Simulation
Simulate Clinical Studies Before First Patient In
Floyd Study Simulation enables research teams to model how a clinical study may behave before or during execution. Using AI-generated virtual participants, TrialKit AI can generate synthetic datasets that mirror the structure of the actual study design.
These simulations allow teams to:
- Evaluate protocol assumptions
- Test inclusion and exclusion criteria
- Explore treatment arm performance
- Forecast event accumulation
- Assess endpoint behavior
Complex multi-year study scenarios can be simulated and analyzed in hours rather than weeks, helping research teams refine study strategies before first patient in.
Floyd Validation
Validate Protocol Design & Study Configuration
Study errors and protocol issues often emerge after a study is already underway. Floyd Validation helps research teams identify potential issues earlier by evaluating study configuration, CRF structure, and protocol logic before study startup.
This capability allows organizations to:
- Generate CRFs
- Validate CRF design
- Identify protocol inconsistencies
- Confirm data structure alignment
- Evaluate study configuration before go-live
By validating study design earlier, research teams can reduce costly mid-study adjustments and improve overall study quality.
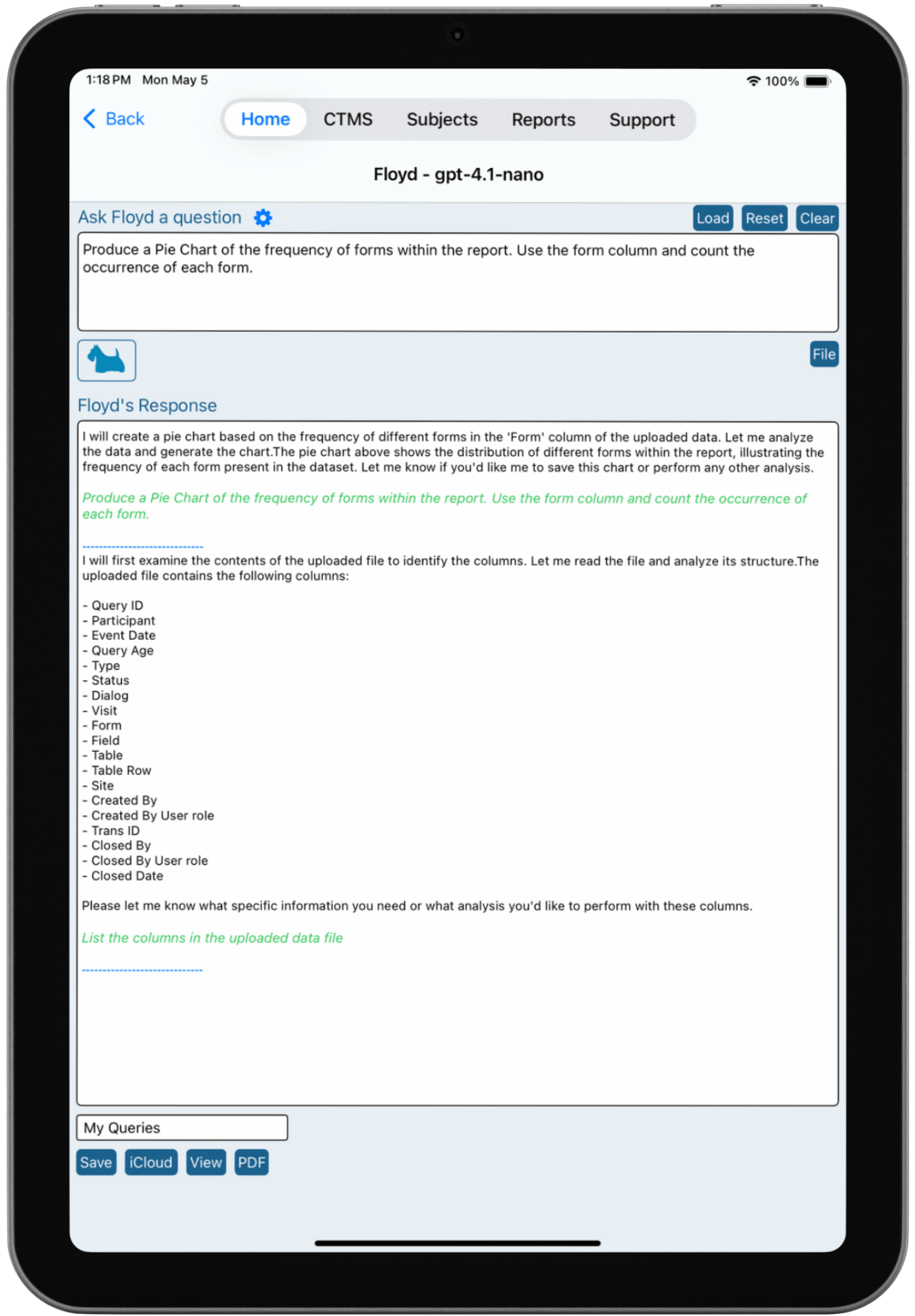
Floyd Chat
Ask Questions. Get Answers Instantly.
Floyd Chat allows clinical research teams to interact with their study data using natural language. Instead of building reports or writing complex queries, users can simply ask questions such as:
- Which participants experienced an adverse event within 14 days of dosing?
- What is the current screen failure rate by site?
- How does enrollment compare to projected timelines?
Floyd instantly analyzes the underlying dataset and returns visual outputs, statistical summaries, and contextual explanations. Floyd can also suggest ways to optimize protocol design and even generate draft protocols based on study requirements, supporting earlier decision-making during study planning.
This allows research teams to investigate questions in seconds rather than days.
Floyd Analytics
Advanced Statistical Analysis in Real Time
Floyd Analytics performs complex biostatistical analysis directly within the TrialKit platform. Research teams can evaluate:
- Treatment efficacy across study arms
- Adverse event trends
- Enrollment and site performance
- Endpoint behavior
- Longitudinal patient outcomes
Statistical outputs such as p-values, confidence intervals, and comparative analyses can be generated instantly, enabling teams to explore study performance without relying on external statistical environments.
This enables continuous analysis across study phases, including interim review and study closeout.
TrialKit AI Timeline Value
What if study setup didn’t take months? This comparison breaks down each step of a traditional workflow against an AI-enabled approach, revealing where timelines are dramatically reduced.

A Unified Platform with Embedded Intelligence
TrialKit is designed as a unified eClinical platform supporting the full lifecycle of clinical trials and non-interventional research. The platform includes:
- EDC
- eSource
- eCOA / ePRO
- eConsent
- eTMF
- RTSM
- Medical Coding
- PACS/Imaging
- Adjudication
- Virtual visits and scheduling
- Remote patient monitoring and wearable connectivity
- Analytics and reporting
TrialKit AI extends these capabilities with embedded intelligence powered by Floyd. Because these capabilities operate within the same platform where study data is captured and managed, research teams can design, simulate, validate, analyze, and interpret studies without exporting datasets or relying on separate tools.
This unified approach allows organizations to both execute and understand their clinical trials within one connected system.
Why Choose TrialKit AI for Your Clinical Trials?
Clinical trials span multiple stages, from design and study build through validation and analysis. TrialKit AI supports each step within a unified platform, helping research teams move faster, evaluate decisions earlier, and better understand how studies will perform.
Accelerate study setup & configuration
Reduce time required for protocol development, form creation, and database build through AI-assisted generation and validation.
Validate study design early
Identify issues in protocol configuration, CRF structure, and study setup before study startup. Reduce the need for mid-study adjustments and improve overall study quality.
Enable simulation-driven study planning
Model full study scenarios using synthetic participant populations that reflect real-world variability. Evaluate protocol assumptions, inclusion criteria, and endpoint behavior before execution begins.
Analyze data without delay
Perform advanced statistical analysis directly within the platform. Assess treatment arms, identify trends, and generate insights without waiting on external reporting workflows.
Work within a single unified platform
All capabilities operate within the TrialKit environment used to design and manage studies. There is no need to export data or rely on separate systems for analysis or simulation.
Support more efficient, informed trials
By enabling earlier evaluation and faster analysis, TrialKit AI helps teams reduce uncertainty, improve decision-making, and execute studies with greater confidence.
Get Started with TrialKit AI Today
Explore how TrialKit AI helps research teams simulate studies, analyze data, and uncover insights faster within the unified TrialKit platform.